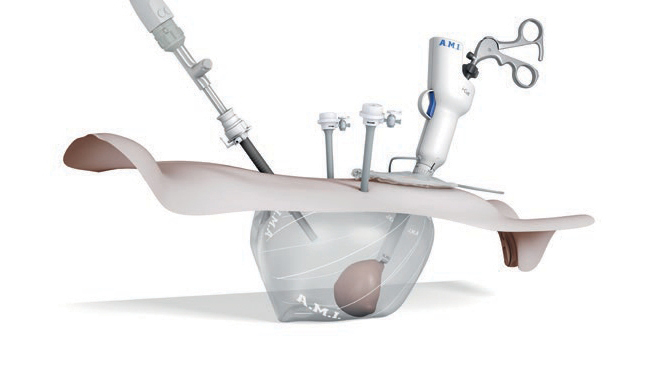
More-Cell-Safe is one of the first solutions available on the market that significantly reduces the risk of unexpected cancer cell scatter during laparoscopic morcellation. Small pieces of tissue, cell particles and fluids, resulting from morcellation, are isolated in a closed, containment system.
Key Features and Benefits
- Elastic, cell- and fluid tight material, which can be insufflated with CO2
- Transparent bag allows clear visualization of surrounding tissue during the procedure
- Dual opening design allows separate insertion of morcellator and optic
- Visi-Shield is a special sleeve used to protect the optic from contamination. After removal, the non-contaminated optic can be used for the remaining operation
- Suitable for use with the Moresolution Morcellator and other types of power morcellators
- Minimal-access surgery which is feasible even for very large uteri (up to 1400 g)
- Minimizes the risk of inadvertent tissue dissemination
- Very low risk of intraoperative complications